Interpret the ECG
shown in Figure-1 — obtained
from a patient with new-onset chest
pain. There is an obvious acute STEMI (ST Elevation Myocardial Infarction).
Follow-up ECGs on this patient are shown in Figure-2 (obtained a
short while later) — and finally in Figure-3 (obtained
post-cath/reperfusion).
- Is there evolution of the MI on these serial
ECGs? What are the specific changes
you see as you compare these sequential tracings?
- Which coronary artery is likely to be acutely occluded?
- Was acute reperfusion successful (Figure-3)?
 |
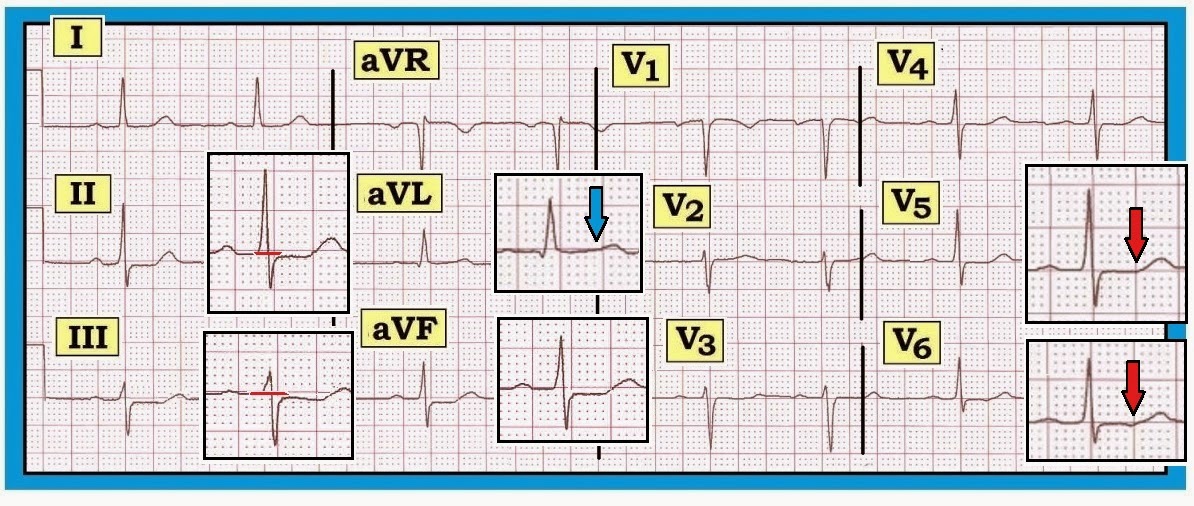
| Figure-1: This is ECG #1 (blue border) — from this patient with new-onset chest pain. There is sinus bradycardia with marked precordial ST elevation. Q waves have not yet formed in the anterior leads on this initial ECG #1. Note the hyperacute appearance of ST-T waves in leads V2,V3,V4. Surprisingly — reciprocal changes are minimal (no more than slight ST-T wave flattening/depression in the inferior leads). Despite this — there can be little doubt that this ECG #1 represents a large acute STEMI in evolution. |
------------------------------------------
What
is the “Culprit” Artery?: We suspect acute proximal LAD occlusion as the “culprit” artery for the acute STEMI seen in Figure‑1. This
is suggested by the ECG finding of diffuse
precordial ST elevation that is especially marked
in leads V2-to-V4.
- Acute occlusion of the LMain (Left Main) Coronary Artery
is rarely seen in practice — because it usually leads to rapid demise of the
patient. In addition to its uncommon occurrence — another clue that the ECG in Figure-1 does not represent LMain occlusion is
that ST elevation is clearly more
marked in lead V1 than in lead aVR. In contrast, with LMain disease or
occlusion — ST elevation is generally more marked in aVR compared to V1.
- This patient is an ideal candidate for acute reperfusion — because there is marked ST elevation in Figure-1,
but no anterior Q waves have yet
formed. The cath lab should be immediately
activated.
Two follow-up ECGs to Figure‑1 are shown
below. For clarity — We use a different
color border for each tracing:
- Figure-1 — ECG
#1 (blue border) = the initial
ECG obtained at presentation.
- Figure-2 — ECG
#2 (red border) =
obtained a short while after ECG #1.
- Figure-3 — ECG
#3 (green border) =
obtained after acute cath and angioplasty/stenting of the acutely occluded LAD.
As you evaluate these serial
ECGs — Keep in mind the following
Questions:
- Is there ECG evidence of evolution on these serial ECGs?
- Was acute reperfusion successful (Figure-3)?
 |
| Figure-2: This is ECG #2 (red border) — obtained a short while after ECG #1 from this patient with acute STEMI. Note that since ECG #1 — there has been interim development of RBBB (an rSr’ complex is now seen in V1 with wide terminal S waves in leads I,V6). The appearance of lead V2 is concerning — as the large new Q wave and now T wave inversion in this lead suggest ongoing evolution is in progress. |
------------------------------------------
 |
| Figure-3: This is ECG #3 (green border) — obtained after acute catheterization and angioplasty/stenting of the acutely occluded LAD. The “good news” — is that this post-cath ECG #3 is encouraging! Note that the QRS complex has narrowed and RBBB is no longer present. The Q wave seen earlier in lead V2 of ECG #2 has resolved — and ST-T waves have essentially returned to baseline. R wave progression is essentially normal (with transition between V3-to-V4). It appears that acute reperfusion has salvaged significant myocardium! |
------------------------------------------
BOTTOM
Line: Use of serial ECGs may be extremely valuable in
following the course of acute MI. Lead-to-lead comparison of QRS
morphology and ST‑T wave changes facilitates determining which changes are new — as well as providing insight to the likely
benefit obtained from acute
intervention.
------------------------------------------
The LAD: Taking A Closer Look
The normal (expected) coronary anatomy of the LCA (Left-Coronary
Artery) is depicted in schematic
Figure-4:
- The LCA arises from the left aortic sinus. This vessel begins as the LMain (Left Main Coronary Artery), which is typically a
short vessel (<10mm) that then bifurcates
into the LAD (Left Anterior Descending Artery) and
the LCx (Left Circumflex Coronary Artery).
Major
Branches of the LAD: The LAD (Left-Anterior-Descending) Artery runs along the
anterior epicardial surface of the heart in the interventricular groove on its
path toward the cardiac apex. The LAD generally supplies the anterior wall of the heart, the cardiac apex and a major portion of the conduction system.
- The major branches of the LAD are i) the Septal perforator vessels; and ii) Diagonal branches.
- Septal branch anatomy is highly variable. We show 2 septal branches in Figure-4
(S-1; S-2) — but instead there may be only one septal branch or
many septal branches, depending on individual
anatomy. The 1st septal
branch is typically the largest; its takeoff is
generally just after the takeoff of the
1st diagonal branch.
- The interventricular septum is the most densely vascularized area of
the heart. This is as it should be given the integral role of the septum in
providing blood supply to the heart’s conduction system. Septal perforators normally run a
vertical path downward following their takeoff
from the proximal LAD.
- Downward penetrating septal branches from the LAD typically connect
with upward penetrating septal
branches from the PDA branch of the RCA. In this way — there is usually a network of collaterals from both LCA and RCA systems in the event of disease in one system.
How adequately collaterals from one system compensate for disease in the other
is subject to individual variation (as well as to how rapidly
occlusive disease develops).
- Clinical Note: Very proximal LAD lesions have been known as “widow-makers”. Especially if
proximal to the 1st septal perforator (and
the 1st diagonal branch) — these
lesions are virtual “left-main-equivalents” because of the extent of injury and
conduction system damage they cause.
- Diagonal branch anatomy is also highly variable. We show 2 diagonal branches in Figure-4
(D-1; D-2) — but there may be 1, 2, or 3 diagonal branches supplying
the anterolateral wall of
the heart. Occasionally — there is no diagonal branch per se, but rather a discrete ramus intermedius arising from between the LAD and LCx to supply the anterolateral surface (not
shown on Figure-4). Typically — it is the 1st diagonal
branch that is the largest.
- Clinical Note: Considerable variation in number and
course of diagonal branch anatomy (and
the angulated path that these vessels follow) may require multiple views on
cath to determine if occlusion is present.
- NOTE-2: Additional variations in anatomy are
not uncommon. One to be aware of is a “wraparound” LAD — in which the LAD is a larger and longer vessel, to the point of extending beyond the cardiac apex and “wrapping
around” to supply the undersurface
(= inferior wall) of the heart.
Awareness of this anatomic variant provides one explanation for the ECG pattern
of simultaneous ST elevation in inferior and anterior lead areas that may sometimes be seen due to acute
occlusion of a single vessel.
 |
| Figure-4: Normal coronary anatomy of the left coronary artery and its major branches. The LCA (Left Coronary Artery) begins as a short LMain (Left Main Coronary Artery) branch — which then bifurcates into the LAD (Left Anterior Descending Artery) and the LCx (Left Circumflex Artery). Panel A — anterior view. Panel B — RAO (Right-Anterior Oblique) view. Abbreviations: S‑1,S‑2 (Septal Perforator branches); D‑1,D‑2 (Diagonal branches); M‑1,M‑2 (Obtuse Marginal branches from the LCx). |
------------------------------------------
Acute
LAD Occlusion:
ECG findings arising from acute LAD occlusion may vary depending on: i) The relative site of
occlusion within the LAD (ie, proximal to
septal perforators and the 1st diagonal or more distal occlusion); ii)
Any prior infarctions that may have
occurred; iii) Presence of any
anatomic variants (such as a
“wrap-around” LAD circulation); and iv) The status of the collateral
circulation. For simplicity — our comments below relate to expected ECG findings assuming no
prior infarctions; no alteration in
collateral circulation; and no
anatomic variants.
- Acute
LAD occlusion
leads to acute anterior MI. This may be extensive and also involve the lateral wall.
- The most typical ECG manifestation of acute LAD occlusion is ST elevation in anterior leads (usually in ≥2 leads between V1-to-V4).
PEARL: ST
elevation in lead aVL
— may provide an invaluable clue to
the location of the acutely occluded coronary artery. According to a study by
Birnbaum et al (Am Heart J 131:38, 1996):
- Suspect acute LAD occlusion proximal to the 1st Diagonal IF in
addition to ST elevation in aVL — there is also ST elevation in leads V2-through-V5. This is the most common
situation when there is ST elevation in lead aVL.
- Suspect 1st
Diagonal branch occlusion IF
in addition to ST elevation in aVL —
there is ST elevation in lead V2 (but not
in V3,V4,V5).
- Suspect LCx
occlusion (especially of the 1st obtuse marginal branch) — IF there is ST elevation in aVL but not in lead V2 (and not in other anterior leads).
NOTE: Anterior
ST elevation without ST elevation in lead aVL — usually suggests more distal LAD occlusion after
takeoff of the 1st Diagonal.
- PEARL: In addition to recognizing ST
elevation in lead aVL with marked
anterior ST elevation — there are 2 additional ways to identify patients at high
risk of impending proximal
LAD occlusion. These are: i)
Recognition of Wellens’ Syndrome (Click here for more on Wellens' Syndrome); and ii) Recognition of DeWinter T Waves (See ECG Blog #53).
------------------------------------------
RETURN
to Figure-1: Is this Proximal LAD
Occlusion?
Application of the above concepts to the ECG shown in Figure-1
(reproduced below in Figure-5) — supports our presumption
of a proximal LAD occlusion. Although
this patient “failed to read the textbook”, in that there is no ST elevation in lead aVL — proximal
LAD occlusion is still strongly suggested because: i) There is marked ST elevation in all anterior
leads, including significant ST
elevation in lead V1; ii) ST
elevation in lead V1 is clearly more
than in lead aVR (virtually no ST
elevation in aVR); and, iii)
The patient developed septal Q waves
(in lead V2) as well as RBBB on the follow-up tracing (Figure-2). RBBB
and the septal Q wave fortunately resolved following the good result obtained
from acute reperfusion.
 |
| Figure-5: This is ECG #1 (reproduced from Figure-1) — obtained from this patient with new-onset chest pain. Despite lack of ST elevation in lead aVL — we strongly suspect proximal LAD occlusion (See text). |
-------------------------------------
Link to Section 10.0 for pdf download on the ECG Diagnosis of Acute MI (from our ECG-2014-ePub).
- ECG Changes of Acute MI — begins in Section 10.1 -
- Discussion of the Coronary Circulation (and determining the "culprit" artery) — begins in Section 10.16 -
- See ECG BLOG #80 for a case involving differentiation between acute RCA vs LCx occlusion.
------------------------------------